Abstract
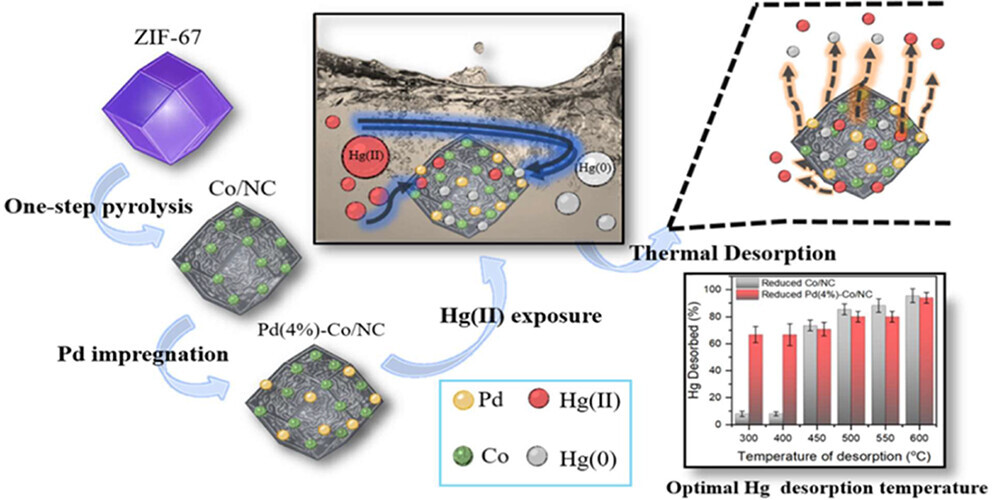
The effectiveness of materials in aqueous contaminant treatment technologies by sorptive removal relies on their ability to be reused and their removal efficiency. Thermal desorption stands out as a promising method to improve the reusability of these materials. In this study, Zeolitic Imidazolate Framework-67 (ZIF-67) derived Cobalt N-Doped Carbon (Co/NC) and metal-impregnated (Ru, Pt, and Pd) Co/NC nanoparticles have been synthesized and tested for the effective removal of aqueous Hg(II) and its reusability by thermal desorption. Reduced Co/NC efficiently removed Hg(II), adsorbing 99.9% of aqueous Hg(II) in 2.5 min through pyridinic-N adsorption sites and Co0 reducing Hg(0) on the surface. The testing of various metals (Ru, Pt, and Pd) on the surface of Co/NC showed that Pd(4%)-Co/NC achieved the highest reactivity with a maximum adsorption capacity of 49.93 mgg–1 using the Langmuir model. Pd(8%)-Co/NC showed the highest adsorbed Hg(0) (79.1%) and fastest removal kinetics (135.52 g mg–1 min–1). The Pd(4%)-Co/NC catalyst retained its durability and stability, eliminating 99.9% of the aqueous Hg species throughout 10 consecutive cycles. The 80.11% and 85.4% of adsorbed Hg were recovered by thermal desorption at 500 °C on Pd(4%)-Co/NC and Co/NC surfaces, respectively. Pd(4%)-Co/NC displayed notable promise as a sustainable catalyst for Hg(II) reductive removal in wastewater treatment technologies, emphasizing its enduring effectiveness and reuse potential for practical engineering applications.
Acknowledgments
This research has been supported by research grants from the Ministry of Science and Higher Education of the Republic of Kazakhstan (BR24993138 and AP14870836) and Nazarbayev University (211123CRP1605). The authors would like to thank the anonymous reviewers who helped significantly improve the quality of the paper.